The FDA on October 21, 2022, approved IMJUDO® (Tremelimumab) in combination with IMFINZI® (Durvalumab) for adult patients with unresectable HepatoCellular Carcinoma (uHCC). IMJUDO® is a product of AstraZeneca Pharmaceuticals.
Tag: Liver Cancer (Hepatocellular)
FDA Approves IMJUDO® plus IMFINZI® for Patients with Advanced Hepatocellular Carcinoma
SUMMARY: The FDA on October 21, 2022, approved IMJUDO® (Tremelimumab) in combination with IMFINZI® (Durvalumab), for adult patients with unresectable HepatoCellular Carcinoma (HCC). The American Cancer Society estimates that for 2022, about 41,260 new cases of primary liver cancer and intrahepatic bile duct cancer will be diagnosed in the US and 30,520 patients will die of their disease. Liver cancer is seen more often in men than in women and the incidence has more than tripled since 1980. This increase has been attributed to the higher rate of Hepatitis C Virus (HCV) infection among baby boomers (born between 1945 through 1965). Obesity and Type II diabetes have also likely contributed to the increasing trend. Other risk factors include alcohol, which increases liver cancer risk by about 10% per drink per day, and tobacco use, which increases liver cancer risk by approximately 50%. HepatoCellular Carcinoma (HCC) is also a leading cause of cancer deaths worldwide, accounting for more than 700,000 deaths each year, and majority of patients typically present at an advanced stage. The prognosis for unresectable HCC remains poor and one year survival rate is less than 50% following diagnosis and only 7% of patients with advanced disease survive five years. NEXAVAR® was approved by the FDA in 2007 for the first line treatment of unresectable HepatoCellular Carcinoma (HCC) and the median Overall Survival was 10.7 months in the NEXAVAR® group and 7.9 months in the placebo group.
Immune checkpoints are cell surface inhibitory proteins/receptors that are expressed on activated T cells. They harness the immune system and prevent uncontrolled immune reactions by switching off the T cells of the immune system. Immune checkpoint proteins/receptors include CTLA-4 (Cytotoxic T-Lymphocyte Antigen 4, also known as CD152) and PD-1(Programmed cell Death 1). Checkpoint inhibitors unleash the T cells resulting in T cell proliferation, activation, and a therapeutic response.
IMFINZI® (Durvalumab) is a human monoclonal antibody that binds to the PD-L1 protein and blocks the interaction of PD-L1 with the PD-1 and CD80 proteins, countering the tumor’s immune-evading tactics and unleashes the T cells. IMJUDO® is a human monoclonal antibody that targets and blocks the activity of CTLA-4, contributing to T-cell activation, priming the immune response to cancer and fostering cancer cell death. In a Phase II study, a single priming dose of IMJUDO® added to IMFINZI® (STRIDE regimen), showed encouraging clinical activity and limited toxicity in patients with unresectable HepatoCellular Carcinoma (HCC), suggesting that a single exposure to IMJUDO® may be sufficient to improve upon activity of IMFINZI®.
HIMALAYA trial is a randomized, open-label, multicenter, global, Phase III study conducted in 190 centres across 16 countries, including in the US, Canada, Europe, South America and Asia. In this study, 1,171 patients with Stage III or IV unresectable HepatoCellular Carcinoma who had received no prior systemic therapy and were not eligible for locoregional therapy (treatment localized to the liver and surrounding tissue), were randomly assigned to receive either the STRIDE regimen which consisted of a single priming dose of IMJUDO® 300 mg IV added to IMFINZI® (Durvalumab) 1500 mg IV, followed IMFINZI® 1500 mg IV by every 4 weeks (N= 393), IMFINZI® monotherapy given at the same dose and schedule (N = 389) or NEXAVAR® (Sorafenib) 400 mg orally BID (N=389). Enrolled patients had ECOG performance status of 0 or 1 and Child-Pugh A disease and could not have main portal vein thrombosis. Patients were stratified based on macrovascular invasion (Yes versus No), etiology of liver disease (Hepatitis B virus versus Hepatitis C virus versus others), and ECOG Performance Status (0 versus 1). The Primary endpoint was Overall Survival (OS) for STRIDE regimen versus NEXAVAR® and key Secondary endpoints included OS for IMFINZI® monotherapy versus NEXAVAR®, Objective Response Rate and Progression Free Survival (PFS) for STRIDE and IMFINZI® monotherapy. The present FDA approval was based on a comparison of the 782 patients randomized to IMJUDO® plus IMFINZI® (STRIDE regimen) to NEXAVAR®.
The Primary objective of this study was met at the time of data cutoff. At a median follow up of 16.1 months of treatment with the STRIDE regimen, there was a 22% reduction in the risk of death for patients who received the STRIDE regimen compared to NEXAVAR® alone (HR=0.78; P=0.0035). The median OS with the STRIDE regimen was 16.4 months, compared with 13.8 months with NEXAVAR®, and the 3 year OS rate was 30.7% versus 20.2 % respectively. The Overall Response Rate for the combination STRIDE regimen was 20.1% compared to 5.1% for NEXAVAR®.
IMFINZI® monotherapy met the objective of OS non-Inferiority to NEXAVAR® (HR=0.86), and the median OS after 16.5 months of median follow up was 16.6 months with IMFINZI® monotherapy versus 13.8 months with NEXAVAR®, and the 3 year OS rate was 24.7% versus 20.2 % respectively. The Overall Response Rate with IMFINZI® monotherapy was 17% compared to 5.1% for NEXAVAR®. The Secondary endpoint of PFS was not superior in either investigational study group relative to the NEXAVAR® control arm. The most common adverse reactions occurring in patients were rash, diarrhea, fatigue, pruritis, musculoskeletal pain and abdominal pain.
It was concluded that HIMALAYA is the first large Phase III trial to add a novel single priming dose of an anti-CTLA4 antibody IMJUDO®, to another checkpoint inhibitor, IMFINZI®. This combination regimen (STRIDE) demonstrated superior efficacy and a favorable benefit-risk profile when compared with NEXAVAR® and should be considered a novel , first-line standard of care systemic therapy, for patients with unresectable Hepatocellular Carcinoma.
Tremelimumab plus Durvalumab in Unresectable Hepatocellular Carcinoma. Abou-Alfa GK, Lau G, Kudo M, et al. Published June 6, 2022. NEJM Evid 2022; 1 (8) DOI:https://doi.org/10.1056/EVIDoa2100070.
IMFINZI® Plus Tremelimumab Significantly Improves Overall Survival in Advanced Hepatocellular Carcinoma
SUMMARY: The American Cancer Society estimates that for 2022, about 41,260 new cases of primary liver cancer and intrahepatic bile duct cancer will be diagnosed in the US and 30,520 patients will die of their disease. Liver cancer is seen more often in men than in women and the incidence has more than tripled since 1980. This increase has been attributed to the higher rate of Hepatitis C Virus (HCV) infection among baby boomers (born between 1945 through 1965). Obesity and Type II diabetes have also likely contributed to the increasing trend. Other risk factors include alcohol, which increases liver cancer risk by about 10% per drink per day, and tobacco use, which increases liver cancer risk by approximately 50%. HepatoCellular Carcinoma (HCC) is also a leading cause of cancer deaths worldwide, accounting for more than 700,000 deaths each year, and majority of patients typically present at an advanced stage. The prognosis for unresectable HCC remains poor and one year survival rate is less than 50% following diagnosis and only 7% of patients with advanced disease survive five years. NEXAVAR® was approved by the FDA in 2007 for the first line treatment of unresectable HepatoCellular Carcinoma (HCC) and the median Overall Survival was 10.7 months in the NEXAVAR® group and 7.9 months in the placebo group.
Immune checkpoints are cell surface inhibitory proteins/receptors that are expressed on activated T cells. They harness the immune system and prevent uncontrolled immune reactions by switching off the T cells of the immune system. Immune checkpoint proteins/receptors include CTLA-4 (Cytotoxic T-Lymphocyte Antigen 4, also known as CD152) and PD-1(Programmed cell Death 1). Checkpoint inhibitors unleash the T cells resulting in T cell proliferation, activation, and a therapeutic response.
IMFINZI® (Durvalumab) is a human monoclonal antibody that binds to the PD-L1 protein and blocks the interaction of PD-L1 with the PD-1 and CD80 proteins, countering the tumor’s immune-evading tactics and unleashes the T cells. Tremelimumab is a human monoclonal antibody that targets and blocks the activity of CTLA-4, contributing to T-cell activation, priming the immune response to cancer and fostering cancer cell death. In a Phase II study, a single priming dose of Tremelimumab added to IMFINZI® (STRIDE regimen), showed encouraging clinical activity and limited toxicity in patients with unresectable HepatoCellular Carcinoma (HCC), suggesting that a single exposure to Tremelimumab may be sufficient to improve upon activity of IMFINZI®.
HIMALAYA trial is a randomized, open-label, multicentre, global, Phase III study conducted in 190 centres across 16 countries, including in the US, Canada, Europe, South America and Asia. In this study, 1,171 patients with Stage III or IV unresectable hepatocellular carcinoma who had received no prior systemic therapy and were not eligible for locoregional therapy (treatment localized to the liver and surrounding tissue), were randomly assigned to receive either the STRIDE regimen which consisted of a single priming dose of Tremelimumab 300 mg IV added to IMFINZI® (Durvalumab) 1500 mg IV, followed IMFINZI® 1500 mg IV by every 4 weeks (N= 393), IMFINZI® monotherapy given at the same dose and schedule (N = 389) or NEXAVAR® (Sorafenib) 400 mg orally BID (N=389). Enrolled patients had ECOG performance status of 0 or 1 and Child-Pugh A disease and could not have main portal vein thrombosis. Patients were stratified based on macrovascular invasion (Yes versus No), etiology of liver disease (Hepatitis B virus versus Hepatitis C virus versus others), and ECOG Performance Status (0 versus 1). The Primary endpoint was Overall Survival (OS) for STRIDE regimen versus NEXAVAR® and key Secondary endpoints included OS for IMFINZI® monotherapy versus NEXAVAR®, Objective Response Rate and Progression Free Survival (PFS) for STRIDE and IMFINZI® monotherapy.
The Primary objective of this study was met at the time of data cutoff. At a median follow up of 16.1 months of treatment with the STRIDE regimen, there was a 22% reduction in the risk of death for patients who received the STRIDE regimen compared to NEXAVAR® alone (HR=0.78; P=0.0035). The median OS with the STRIDE regimen was 16.4 months, compared with 13.8 months with NEXAVAR®, and the 3 year OS rate was 30.7% versus 20.2 % respectively. The Overall Response Rate for the combination STRIDE regimen was 20.1% compared to 5.1% for NEXAVAR®
IMFINZI® monotherapy met the objective of OS non-Inferiority to NEXAVAR® (HR=0.86; 96% CI, 0.73–1.03) and the median OS after 16.5 months of median follow up was 16.6 months with IMFINZI® monotherapy versus 13.8 months with NEXAVAR®, and the 3 year OS rate was 24.7% versus 20.2 % respectively. The Overall Response Rate with IMFINZI® monotherapy was 17% compared to 5.1% for NEXAVAR®. The Secondary endpoint of PFS was not superior in either investigational study group relative to the NEXAVAR® control arm.
It was concluded that HIMALAYA is the first large Phase III trial to add a novel single priming dose of an anti-CTLA4 antibody Tremelimumab, to another checkpoint inhibitor, IMFINZI®. This combination regimen (STRIDE) demonstrated superior efficacy and a favorable benefit-risk profile when compared with NEXAVAR® and should be considered a novel , first-line standard of care systemic therapy, for patients with unresectable Hepatocellular Carcinoma.
Phase 3 randomized, open-label, multicenter study of tremelimumab (T) and durvalumab (D) as first-line therapy in patients (pts) with unresectable hepatocellular carcinoma (uHCC): HIMALAYA. Abou-Alfa GK, Chan SL, Kudo M, et al. J Clin Oncol. 2022;40(suppl 4):379. doi:10.1200/JCO.2022.40.4_suppl.379
FDA Approves TheraSphere Y-90 Glass Microspheres for Hepatocellular Carcinoma
SUMMARY: The FDA on March 18, 2021 approved TheraSphere Y-90 Glass Microspheres, developed for the treatment of patients with HepatoCellular Carcinoma (HCC). The American Cancer Society estimates that for 2021, about 42,230 new cases of primary liver cancer and and intrahepatic bile duct cancer will be diagnosed in the US and 30,230 patients will die of their disease. Liver cancer is seen more often in men than in women and the incidence has more than tripled since 1980. This increase has been attributed to the higher rate of Hepatitis C Virus (HCV) infection among baby boomers (born between 1945 through 1965). Obesity and Type II diabetes have also likely contributed to the increasing trend. Other risk factors include alcohol, which increases liver cancer risk by about 10% per drink per day, and tobacco use, which increases liver cancer risk by approximately 50%. HepatoCellular Carcinoma (HCC) is the second most common cause of cancer-related deaths worldwide, and majority of patients typically present at an advanced stage. The prognosis for unresectable HCC remains poor and one year survival rate is less than 50% following diagnosis.
Patients with HCC, with disease confined to one lobe of the liver often undergo liver resection if feasible, and liver transplantation when HCC is associated with chronic liver disease and cirrhotic livers. Other liver-directed therapies include ablative modalities such as SBRT (Stereotactic Body Radiation Therapy), Cryoablation, RFA (RadioFrequency Ablation), as well as Trans Arterial ChemoEmbolization (TACE) and Selective Internal Radiation Therapy (SIRT), also referred to as radioembolization. HepatoCellular Carcinoma’s derive their blood supply almost exclusively from the hepatic arteries, whereas the liver has a dual blood supply from the portal vein (75-80%) and hepatic arteries (20-25%). By taking advantage of the differential blood supply of hepatic tumors, chemo and radioembolization can be targeted to the tumor tissue, minimizing damage to the surrounding normal liver parenchyma.
TheraSphere treatment is a type of SIRT with low toxicity, and is comprised of millions of microscopic glass beads containing radioactive yttrium (Y-90), which is delivered to liver tumors via a hepatic arterial catheter, and results in minimal radiation exposure to surrounding normal liver parenchyma.
LEGACY trial is a single-arm, multicenter, retrospective study designed to evaluate the safety and efficacy of Yttrium-90 (Y90) glass microspheres in patients with unresectable solitary hepatocellular carcinoma (HCC). The objective of this study was to assess the Objective Response Rate (ORR) and Duration of Response (DoR) following treatment with Yttrium-90 (Y90), and the objective of the analyses presented was to evaluate ORR, DoR, and Overall Survival (OS) by transplant/resection status (neoadjuvant therapy with the intent to bridge patients to transplant or resection), and to compare these outcomes with patients who did not go on to transplantation/resection after receiving treatment with Y90. This study included 162 patients with Child-Pugh A and Barcelona Clinic Liver Cancer (BCLC) A or C disease, with unresectable solitary liver lesion that was 2-8 cm. The median patient age was 66 yrs and patients with portal vein thrombosis or extrahepatic disease were excluded. No prior liver transplantation, resection, locoregional treatment or systemic therapy was allowed. Primary efficacy endpoints included Objective Response Rate (ORR) and Duration of Response (DoR). ORR included patients who achieved either a Complete response or Partial Response, and response was determined by Blinded Independent Central Review (BICR). Secondary endpoints include Overall Survival (OS) and number and type of subsequent treatments, including transplantation and resection.
This study met both Primary endpoints and the ORR was 72.2% at 4 weeks and DoR (more than 6 months) was 76.1%. The median follow up for all 162 patients enrolled was 29.9 months and 3-year OS was 86.6%. Among the 45 of the total 162 patients who received neoadjuvant Y90 treatment, 21% went on to transplantation and 7% went on to have resection. In the neoadjuvant treatment group, ORR was 100%, DoR (more than 6 months) was 30.6% and 3-year OS was 93%. Liver function as determined by levels of albumin and bilirubin were maintained in over 85% of patients.
It was concluded that treatment of solitary unresectable HCC with Y90 glass microspheres, performed as an outpatient procedure, resulted in significant ORR, DoR, and OS, both as neoadjuvant therapy prior to transplantation/resection and as treatment in non-surgical candidates.
Use of yttrium-90 (Y90) glass microspheres (TheraSphere) as neoadjuvant to transplantation/resection in hepatocellular carcinoma: Analyses from the LEGACY study. Lewandowski R, Johnson GE, Kim E, et al. DOI: 10.1200/JCO.2021.39.3_suppl.300 Journal of Clinical Oncology 39, no. 3_suppl (January 20, 2021) 300-300.
FDA Approves TECENTRIQ® in Combination with AVASTIN® for Hepatocellular Carcinoma
SUMMARY: The FDA on May 29, 2020, approved TECENTRIQ® (Atezolizumab) in combination with AVASTIN® (Bevacizumab) for patients with unresectable or metastatic HepatoCellular Carcinoma (HCC), who have not received prior systemic therapy. The American Cancer Society estimates that for 2020, about 42,810 new cases of primary liver cancer will be diagnosed in the US and 30,160 patients will die of their disease. Liver cancer is seen more often in men than in women and the incidence has more than tripled since 1980. This increase has been attributed to the higher rate of Hepatitis C Virus (HCV) infection among baby boomers (born between 1945 through 1965). Obesity and Type II diabetes have also likely contributed to the increasing trend. Other risk factors include alcohol, which increases liver cancer risk by about 10% per drink per day, and tobacco use, which increases liver cancer risk by approximately 50%. HepatoCellular Carcinoma (HCC) is the second most common cause of cancer-related deaths worldwide, and majority of patients typically present at an advanced stage. The prognosis for unresectable HCC remains poor and one year survival rate is less than 50% following diagnosis. NEXAVAR® was approved by the FDA in 2007 for the first line treatment of unresectable HepatoCellular Carcinoma (HCC) and the median Overall Survival was 10.7 months in the NEXAVAR® group and 7.9 months in the placebo group.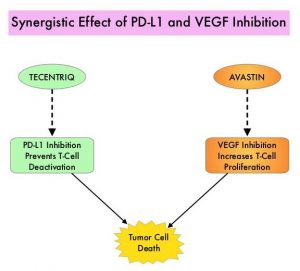
TECENTRIQ® (Atezolizumab) is an anti PD-L1 monoclonal antibody, designed to directly bind to PD-L1 expressed on tumor cells and tumor-infiltrating immune cells, thereby blocking its interactions with PD-1 and B7.1 receptors. PD-L1 inhibition may prevent T-cell deactivation and further enable the activation of T cells. AVASTIN® (Bevacizumab) is a recombinant humanized monoclonal IgG1 antibody that binds VEGF (Vascular Endothelial Growth Factor) and prevents the interaction of VEGF to its receptors (Flt-1 and KDR) on the surface of endothelial cells, thereby preventing endothelial cell proliferation and new blood vessel formation. AVASTIN® in addition to its established anti-angiogenic properties can further enhance TECENTRIQ®’s ability to restore anti-cancer immunity, by inhibiting VEGF-related immunosuppression, promoting T-cell tumor infiltration and enabling priming and activation of T-cell responses against tumor antigens. The use of TECENTRIQ® in combination with AVASTIN® therefore has a strong scientific rationale, as this combination can potentially enhance the immune system to combat a broad range of malignancies.
IMbrave150 is a global, open-label, multicenter, randomized, Phase III study in which a combination of TECENTRIQ® and AVASTIN® was compared with standard-of-care NEXAVAR®, in patients with previously untreated locally advanced or metastatic HCC. Patients were randomized 2:1 to receive TECENTRIQ® 1200 mg IV on day 1 along with AVASTIN® 15 mg/kg on day 1 of each 21-day cycle (N=336) or NEXAVAR® 400 mg orally twice daily, each day of the 21-day cycle (N=165). Treatment was continued until disease progression or unacceptable toxicity. The treatment groups were well balanced and enrolled patients had an ECOG performance status of 0 or 1, Child-Pugh Class A disease, and adequate hematologic and end-organ function. The two co-Primary endpoints were Overall Survival (OS) and Progression Free Survival (PFS). The key Secondary endpoints included Overall Response Rate (ORR), Time To Progression (TTP) and Duration of Response (DOR), as well as Patient-Reported Outcomes (PROs), Safety and Pharmacokinetics.
With a median follow up of 8.6 months, the OS was not yet reached in the TECENTRIQ® and AVASTIN® combination group compared with 13.2 months in the NEXAVAR® group (HR=0.58; P=0.0006). The median PFS was 6.8 months versus 4.3 months respectively (HR=0.59; P<0.0001). The ORR was 27% versus 12% (P<0.0001) based on the Independent Review Facility RECIST 1.1 criteria, in favor of the combination regimen. This benefit was seen across clinical subgroups and the combination regimen delayed deterioration of Quality of Life, compared with NEXAVAR®. Grade 3 and 4 Adverse Events were similar and occurred in 57% and 55% of the combination and control arms, respectively.
It was concluded that a combination of TECENTRIQ® and AVASTIN® demonstrated statistically significant and clinically meaningful improvement in both Overall Survival and Progression Free Survival, compared with NEXAVAR®, in treatment naïve patients with unresectable Hepatocelluar Carcinoma. The authors added that this is the first study in 11 years to show an improvement in Overall Survival with a new first line treatment option, compared to NEXAVAR®, and has the potential to be a practice changing treatment in Hepatocellular Carcinoma.
IMbrave150: Efficacy and safety results from a ph III study evaluating atezolizumab (atezo) + bevacizumab (bev) vs sorafenib (Sor) as first treatment (tx) for patients (pts) with unresectable hepatocellular carcinoma (HCC). Cheng A-L, Qin S, Ikeda M, et al. Annals of Oncology, Volume 30, 2019 Supplement 9. LBA3.
TECENTRIQ® (Atezolizumab) plus AVASTIN® (Bevacizumab)
The FDA on May 29, 2020 approved TECENTRIQ® in combination with AVASTIN® for patients with unresectable or metastatic Hepatocellular Carcinoma (HCC) who have not received prior systemic therapy. TECENTRIQ® and AVASTIN® are products of Genentech Inc.
OPDIVO® and YERVOY®
The FDA on March 10, 2020 granted accelerated approval to the combination of OPDIVO® (Nivolumab) and YERVOY® (Ipilimumab) for patients with Hepatocellular Carcinoma (HCC), who have been previously treated with NEXAVAR® (Sorafenib). Both OPDIVO® and YERVOY® are products of Bristol-Myers Squibb Co.
FDA Approves OPDIVO®/YERVOY® Combination for Advanced Hepatocellular Carcinoma
SUMMARY: The FDA on March 10, 2020, granted accelerated approval to the combination of OPDIVI® (Nivolumab) and YERVOY® (Ipilimumab) for patients with HepatoCellular Carcinoma (HCC) who have been previously treated with NEXAVAR® (Sorafenib). The American Cancer Society estimates that for 2020, about 42,810 new cases of primary liver cancer will be diagnosed in the US and 30,160 patients will die of their disease. Liver cancer is seen more often in men than in women, and the incidence has more than tripled since 1980. This increase has been attributed to the higher rate of Hepatitis C Virus (HCV) infection among baby boomers (born between 1945 through 1965). Obesity and Type II diabetes have also likely contributed to the trend. Other risk factors include alcohol, which increases liver cancer risk by about 10% per drink per day, and tobacco use which increases liver cancer risk by approximately 50%. HepatoCellular Carcinoma is the second most common cause of cancer-related deaths worldwide and majority of patients typically present at an advanced stage. The prognosis for unresectable HCC remains poor and one year survival rate is less than 50% following diagnosis. NEXAVAR® was approved by the FDA in 2007 for the first line treatment of unresectable HepatoCellular Carcinoma (HCC) and the median Overall Survival was 10.7 months in the NEXAVAR® group and 7.9 months in the placebo group.
Immune checkpoints are cell surface inhibitory proteins/receptors that are expressed on activated T cells. They harness the immune system and prevent uncontrolled immune reactions by switching off the T cells of the immune system. Immune checkpoint proteins/receptors include CTLA-4 (Cytotoxic T-Lymphocyte Antigen 4, also known as CD152) and PD-1(Programmed cell Death 1). Checkpoint inhibitors unleash the T cells resulting in T cell proliferation, activation, and a therapeutic response.
OPDIVO® (Nivolumab) is a fully human, immunoglobulin G4 monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, thereby undoing PD-1 pathway-mediated inhibition of the immune response and unleashing the T cells. OPDIVO® was approved by the FDA in 2017, for the treatment of HCC, in patients who have been previously treated with NEXAVAR®. YERVOY® is a fully human immunoglobulin G1 monoclonal antibody that blocks Immune checkpoint protein/receptor CTLA-4 (Cytotoxic T-Lymphocyte Antigen 4).
The present FDA approval was based on the CheckMate-040 study, which is an ongoing Phase I/II, open-label, multi-cohort study, investigating OPDIVO® or OPDIVO®-based combinations in patients with advanced HCC with and without Chronic Viral Hepatitis, who are naïve, intolerant to, or who have progressed during NEXAVAR® therapy. The OPDIVO® plus YERVOY® cohort of CheckMate-040 evaluated the Safety and Efficacy of the combination, in patients with previously treated advanced HCC. This cohort included a total of 49 patients with HCC who progressed on or were intolerant to NEXAVAR®. Patients received OPDIVO® 1 mg/kg IV in combination with YERVOY® 3 mg/kg IV, every 3 weeks for four doses, followed by single agent OPDIVO® 240 mg IV every 2 weeks, until disease progression or unacceptable toxicity. The main efficacy endpoints were Overall Response Rate (ORR) and Duration of Response (DoR), as determined by Blinded Independent Central Review.
At a median follow up of 28 months, the ORR with the OPDIVO® plus YERVOY® combination was 33%, with 8% Complete Responses (CR) and a 24% Partial Responses. The Duration of Responses ranged from 4.6 to over 30.5 months, with 31% of responses lasting at least 24 months. The most common adverse events with this combination therapy were fever, fatigue, diarrhea, rash, arthralgia, dyspnea, and hypothyroidism.
It was concluded that dual Immuno-Oncology therapy with a combination of OPDIVO® plus YERVOY® represents an important milestone, for patients with advanced Hepatocellular Carcinoma. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-nivolumab-and-ipilimumab-combination-hepatocellular-carcinoma
First Line Combination of TECENTRIQ® and AVASTIN® Improves Survival in Hepatocellular Carcinoma
SUMMARY: The American Cancer Society estimates that for 2019, about 42,030 new cases of primary liver cancer will be diagnosed in the US and 31,780 patients will die of their disease. Liver cancer is seen more often in men than in women and the incidence has more than tripled since 1980. This increase has been attributed to the higher rate of Hepatitis C Virus (HCV) infection among baby boomers (born between 1945 through 1965). Obesity and Type II diabetes have also likely contributed to the trend. Other risk factors include alcohol, which increases liver cancer risk by about 10% per drink per day, and tobacco use, which increases liver cancer risk by approximately 50%. HepatoCellular Carcinoma (HCC) is the second most common cause of cancer-related deaths worldwide and majority of patients typically presents at an advanced stage. The prognosis for unresectable HCC remains poor and one year survival rate is less than 50% following diagnosis. NEXAVAR® was approved by the FDA in 2007 for the first line treatment of unresectable HepatoCellular Carcinoma (HCC) and the median Overall Survival was 10.7 months in the NEXAVAR® group and 7.9 months in the placebo group.
TECENTRIQ® (Atezolizumab) is an anti PD-L1 monoclonal antibody, designed to directly bind to PD-L1 expressed on tumor cells and tumor-infiltrating immune cells, thereby blocking its interactions with PD-1 and B7.1 receptors, and thus enabling the activation of T cells. AVASTIN® (Bevacizumab) is a recombinant humanized monoclonal IgG1 antibody that binds VEGF (Vascular Endothelial Growth Factor) and prevents the interaction of VEGF to its receptors (Flt-1 and KDR) on the surface of endothelial cells, thereby preventing endothelial cell proliferation and new blood vessel formation. The use of TECENTRIQ® in combination with AVASTIN® has a strong scientific rationale, as this combination can potentially enhance the immune system to combat a broad range of malignancies. AVASTIN® in addition to its established anti-angiogenic properties can further enhance TECENTRIQ®’s ability to restore anti-cancer immunity, by inhibiting VEGF-related immunosuppression, promoting T-cell tumor infiltration and enabling priming and activation of T-cell responses against tumor antigens.
IMbrave150 is a global, open-label, multicenter, randomized, Phase III study in which a combination of TECENTRIQ® and AVASTIN® was compared with standard-of-care NEXAVAR®, in patients with previously untreated locally advanced or metastatic HCC. Patients were randomized 2:1 to receive TECENTRIQ® 1200 mg IV on day 1 along with AVASTIN® 15 mg/kg on day 1 of each 21-day cycle (N=336) or NEXAVAR® 400 mg orally twice daily, each day of the 21-day cycle (N=165). Treatment was continued until disease progression or unacceptable toxicity. The treatment groups were well balanced and enrolled patients had an ECOG performance status of 0 or 1, Child-Pugh Class A disease, and adequate hematologic and end-organ function. The two co-Primary endpoints were Overall Survival (OS) and Progression Free Survival (PFS). The key Secondary endpoints included Overall Response Rate (ORR), Time To Progression (TTP) and Duration of Response (DOR), as well as Patient-Reported Outcomes (PROs), Safety and Pharmacokinetics.
With a median follow up of 8.6 months, the OS was not yet reached in the TECENTRIQ® and AVASTIN® combination group compared with 13.2 months in the NEXAVAR® group (HR=0.58; P=0.0006). The median PFS was 6.8 months versus 4.3 months respectively (HR=0.59; P<0.0001). The ORR was 27% versus 12% (P<0.0001) based on the Independent Review Facility RECIST 1.1 criteria, in favor of the combination regimen. This benefit was seen across clinical subgroups and the combination regimen delayed deterioration of Quality of Life compared with NEXAVAR®. Grade 3 and 4 Adverse Events were similar and occurred in 57% and 55% of the combination and control arms, respectively.
It was concluded that a combination of TECENTRIQ® and AVASTIN® demonstrated statistically significant and clinically meaningful improvement in both Overall Survival and Progression Free Survival compared with NEXAVAR®, in treatment naïve patients with unresectable Hepatocelluar Carcinoma. The authors added that this is the first study in 11 years to show an improvement in Overall Survival with a new first line treatment option, compared to NEXAVAR®, and has the potential to be a practice changing treatment in Hepatocellular Carcinoma. IMbrave150: Efficacy and safety results from a ph III study evaluating atezolizumab (atezo) + bevacizumab (bev) vs sorafenib (Sor) as first treatment (tx) for patients (pts) with unresectable hepatocellular carcinoma (HCC). Cheng A-L, Qin S, Ikeda M, et al. Annals of Oncology, Volume 30, 2019 Supplement 9. LBA3
CYRAMZA® (Ramucirumab)
The FDA on May 10, 2019 approved CYRAMZA® as a single agent for HepatoCellular Carcinoma (HCC) in patients who have an Alpha FetoProtein (AFP) of 400 ng/mL or more, and have been previously treated with NEXAVAR® (Sorafenib). CYRAMZA® is a product of Eli Lilly and Company.
